UNDERSTANDING OZONE FOR THE LAUNDRY
Ozone gas is used in industry as a laundry booster. It is a highly active energy source that replaces the use of heat in accelerating the work of the water and chemicals in the laundry process, and hastening the destruction of soil and micro-organisms. Ozone gas is a tri-atomic allotrope of oxygen, in other words there are three atoms to an oxygen molecule instead of the normal two.
This gas is unstable and in a wash cycle the extra atom disengages and combines with other chemical molecules, boosting their properties and at the same time oxidising and decomposing soil. Additionally the ozone forms hydroxyl radicals which are 3000 times faster than chlorine at disinfection and 11 times stronger.
The activity of ozone expands the weave of the linen, releasing ingrained soil and chemical re deposition, resulting in improved cleanliness, enhanced whiteness and colours and providing a high level of disinfection.
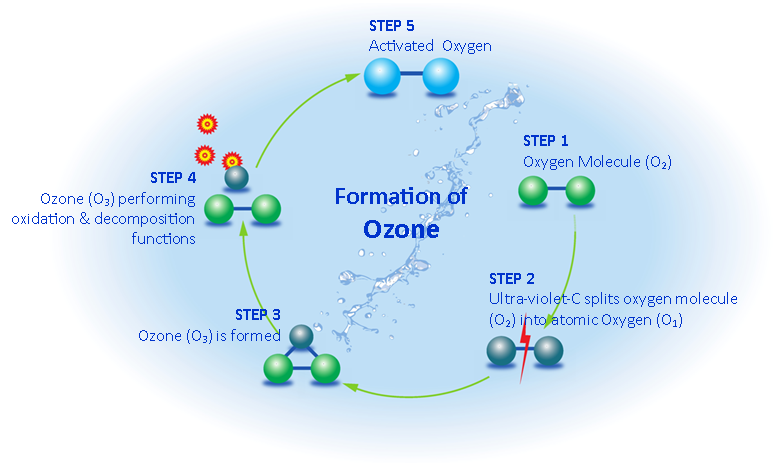
The OTEK ozone laundry system utilises 185 nanometre ultra violet lamps to split the oxygen molecule and form small amounts of ozone. The ozone is then fed into the laundry machine throughout the whole length of the wash.
The formation of ozone gas through U.V lamps as used in the patented Ozone Technologies laun- dry system is a very safe and reliable way of producing ozone gas (see diagram).
IN INDUSTRY, OZONE OR OZONATED WATER IS USED TO:
- Disinfect and boost laundry in hospitals, aged care homes, food factories etc.
- Disinfect water before it is bottled.
- Deodorise air and objects, such as after a fire.
- Kill bacteria on food or on contact surfaces.
- Swimming pool and spa sanitization.
- Scrub yeast and mould spores from the air in food processing plants.
- Wash fresh fruits and vegetables to kill yeast, mould and bacteria.
- Chemically attack contaminants in water (iron, arsenic, hydrogen sulphide, nitrites, and complex organics lumped together as “colour”).
- Provide an aid to flocculation (agglomeration of molecules, which aids in filtration, where the iron and arsenic are removed).
- Manufacture chemical compounds via chemical synthesis.
- Clean and bleach fabrics.
- Assist in processing plastics to allow adhesion of inks.
- Age rubber samples to determine the useful life of a batch of rubber.
- Disinfect hospital operating rooms where air needs to be sterile.
- Eradicate water borne parasites such as Giardia and Cryptosporidium in surface water treatment plants.
- This process is known as Ozonation.
